Impair the cellular interferon response or alternatively the effects of TSA go beyond interferon induced antiviral responses. While we cannot distinguish between these possibilities at the present time, the latter explanation seems likely to us since we and others have found using microarray analysis of HDI treated cells, that hundreds of cellular transcripts induced by viral infection are affected by blocking histone deacetylase activity. Using small molecule OV-enhancers is attractive from a clinical standpoint. In principle, this strategy allows for quite significant genetic attenuation of OVs to restrict growth in normal tissues with conditional rescue of the virus replication in SU5416 tumour cells following treatment with an enhancing compound like an HDI or other classes of molecules that can complement viral defects. In this and our previous studies, one of the key features of small molecule complementation of attenuated viruses is that the effect of virus enhancers is restricted to tumour cells with minimal impact on the anti-viral programs of normal tissues. This clearly is the case for TSA in the studies presented here where we demonstrate both in vitro and in animal models that TSA does not enhance virus growth in normal tissues. The reason for this selectivity is unclear at this time however tumour cells are known to have elevated levels of histone deacetylases suggesting that perhaps malignancies have evolved more dependency upon this type of epigenetic modification to control gene expression. Alternatively since tumour cells have often inactivated at least some components of their anti-viral programs, it may be that the addition of HDIs simply “break the camel��s back” in tumour cells but are ineffective in normal tissues that have fully intact multilayered protection systems. Further studies are currently under way to address some of these issues. We GDC-0879 Raf inhibitor conclude that TSA is a potent enhancer of VV in vitro and in vivo. We propose that HDIs such as TSA could be used to enhance the effectiveness 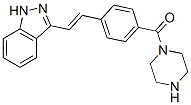 of OVs in vivo and that further clinical evaluation of this possibility is warranted. We therefore reasoned that if the AA target was the cytosolic CyP-A, the drug could also act on other members of this protein family. Indeed, such a pleiotropic effect is well-characterized for CsA, as CsA also targets the mitochondria-restricted CyP-D. CyP-D displays an important role in the cell response to a variety of noxious stimuli, as it modulates a channel located in the inner mitochondrial membrane, the permeability transition pore, whose prolonged opening irreversibly commits cells to death. PTP dysregulation is emerging as a common feature in a variety of pathologies endowed with either an excess of cell death, such as neurodegenerative disease or muscular dystrophies, or with an aberrant hyperactivation of survival pathways, as in cancer. CsA inhibits PTP opening through binding to CyP-D. Therefore, it constitutes an interesting molecule for the treatment of degenerative diseases. Nonetheless, due to its immunosuppressant activity, to its side effects and to its inability to pass the blood-brain barrier, CsA analogues with a higher selectivity for CyP-D are under intense scrutiny. Here we demonstrate that, similar to CsA, AA targets CyP-D leading to PTP inhibition and to cell protection from insults that cause pore opening. AA could be exploited as a lead compound for a new class of PTP-inhibiting drugs. The identification of PTP targeting drugs is a highly desirable result, as the PTP is involved in a wide range of diseases. By the use of CsA or of CyP-D knock-out animals it was established that dysregulated.
of OVs in vivo and that further clinical evaluation of this possibility is warranted. We therefore reasoned that if the AA target was the cytosolic CyP-A, the drug could also act on other members of this protein family. Indeed, such a pleiotropic effect is well-characterized for CsA, as CsA also targets the mitochondria-restricted CyP-D. CyP-D displays an important role in the cell response to a variety of noxious stimuli, as it modulates a channel located in the inner mitochondrial membrane, the permeability transition pore, whose prolonged opening irreversibly commits cells to death. PTP dysregulation is emerging as a common feature in a variety of pathologies endowed with either an excess of cell death, such as neurodegenerative disease or muscular dystrophies, or with an aberrant hyperactivation of survival pathways, as in cancer. CsA inhibits PTP opening through binding to CyP-D. Therefore, it constitutes an interesting molecule for the treatment of degenerative diseases. Nonetheless, due to its immunosuppressant activity, to its side effects and to its inability to pass the blood-brain barrier, CsA analogues with a higher selectivity for CyP-D are under intense scrutiny. Here we demonstrate that, similar to CsA, AA targets CyP-D leading to PTP inhibition and to cell protection from insults that cause pore opening. AA could be exploited as a lead compound for a new class of PTP-inhibiting drugs. The identification of PTP targeting drugs is a highly desirable result, as the PTP is involved in a wide range of diseases. By the use of CsA or of CyP-D knock-out animals it was established that dysregulated.
Monthly Archives: August 2019
During the past several decades many efforts in developing anti influenza drugs due to the rapid mutations
Although some fish AFPs and insect AFPs have shown activity as hydrate inhibitors in propane hydrate, the GFP fusions used here have never been tested, nor have the Torin 1 tested hydrates been formed using a gas mixture that would be found in a high-pressure oil and gas pipeline. Conditions in pipelines can vary, but deep sea lines are generally at 4uC and$1 000 PSI, an environment that we approximated here. These parameters did not seem to have deleterious effects on the proteins because even when the gas hydrates were thawed and the proteins used a second time, inhibition activity was still observed. All of the investigated AFPs showed hydrate inhibition as determined by gas uptake assessments. Similar to the observations on the single THF crystals, LpAFP-GFP and Type III AFP-GFP showed hydrate inhibition that was modestly higher than the chemical inhibitor PVP. GFP showed little inhibition, possibly only due to a colligative effect since, as indicated, no incorporation into THF hydrate was seen. Of the additives tested, Type III AFP was demonstrably superior with an overall 18% decrease in gas hydrate formation, validating again the observations made with this protein on THF single hydrate crystals. In conclusion, we have demonstrated for the first time that AFPs irreversibly adsorb to sII hydrate surfaces and we speculate that they act as inhibitors by binding to the 111 faces of the these symmetrical cubic crystals. We further consider that the identified ice-binding residues of these proteins may not be identical to the residues that interact with the hydrate surface, but the way is now clear for such an investigation. In addition, these experiments have established that AFPs are suitable models for understanding hydrate-inhibitor reactions and offer the prospect that these proteins, or their modified cognates, will be useful as new and more effective biologically-based hydrate inhibitors. Recently, the outbreak of H1N1 influenza A virus is a pandemic of a new strain of influenza virus identified in April 2009, commonly referred to as “swine flu”. Regorafenib Within only four months, the pandemic has caused many deaths from the first detected country Mexico to almost all countries of the world. The H1N1 influenza virus is quite familiar to us because it had caused the 1918�C1919 Spain pandemic that had infected 5% of the world population and resulted in 20�C50 million deaths worldwide. In July 2009 the WHO enhanced the warning to phase 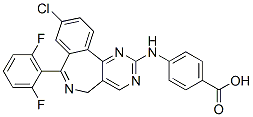 6, meaning that the spread of H1N1 influenza virus has become a serious global pandemic. It was anticipated that a stronger outbreak might occur in the coming winter. The even worse news is that cases were reported that several strains of H1N1 influenza A viruses were resistant to oseltamivir. Although an influenza virus only possesses eight genes, its simplicity has not stopped it from wreaking havoc on human beings for centuries. “The only thing predictable about influenza is its unpredictability”. Influenza A virus has the ability to undergo changes by the mechanisms of antigenic drift and shift, resulting in new evolving virus strains, which may be extremely toxic and drug-resistant. Given that influenza shifts may occur every 20�C30 years, the danger of future influenza A pandemics highlights the need to develop more effective drugs. The threat of an impending influenza pandemic, possibly through the mutations of the present avian strain H5N1 or swine strain H1N1, has triggered a global effort to develop more effective antivirus drugs.
6, meaning that the spread of H1N1 influenza virus has become a serious global pandemic. It was anticipated that a stronger outbreak might occur in the coming winter. The even worse news is that cases were reported that several strains of H1N1 influenza A viruses were resistant to oseltamivir. Although an influenza virus only possesses eight genes, its simplicity has not stopped it from wreaking havoc on human beings for centuries. “The only thing predictable about influenza is its unpredictability”. Influenza A virus has the ability to undergo changes by the mechanisms of antigenic drift and shift, resulting in new evolving virus strains, which may be extremely toxic and drug-resistant. Given that influenza shifts may occur every 20�C30 years, the danger of future influenza A pandemics highlights the need to develop more effective drugs. The threat of an impending influenza pandemic, possibly through the mutations of the present avian strain H5N1 or swine strain H1N1, has triggered a global effort to develop more effective antivirus drugs.
This assumption is corroborated by the observation that OCT1/3mediated metformin uptake into murine hepatocytes
Since OCT1 and OCT3 are expressed in the plasma membrane of human hepatocytes, skeletal muscle cells, and adipocytes, an inhibition potential of OCT function by PPIs may abolish the glucose-lowering effect of metformin. Human adipocytes or human skeletal muscle cells is significantly reduced by known OCT inhibitors such as quinidine or cimetidine. Similarly, the activating effect of metformin on AMP-activated protein kinase is altered. Moreover, certain genotypes may directly affect the inhibition potential of a drug, a mechanism which is increasingly recognized. OCT1 pharmacogenetics and drug-drug interaction for metformin uptake and selected OCT1 inhibitors have recently been reported by in vitro experiments. Interestingly, an increased sensitivity to drug inhibition was observed for OCT variants, particularly for those with reduced function. While the glucose-lowering effect of metformin is LY2109761 impaired either in healthy volunteers or in diabetic Z-VAD-FMK 187389-52-2 patients carrying OCT1 variants with reduced function, no data are currently available with respect to PPI co-medication. Since generally only 60% of metformin-treated patients with type 2 diabetes do respond well, it might be possible that, clinically, PPIs are a yet unrecognized factor for insufficient metformin response due to a transporter-mediated drug-drug interaction via OCT transport proteins. Taken together, we identified PPIs as an important drug class inhibiting OCT-mediated metformin transport. Moreover, our present work underscores the impact of in silico pharmacophore modeling since these computational data have been confirmed by our experimental studies using transfected cell lines, which express functionally active OCTs. Additionally, this newly recognized in vitro drug-drug interaction warrants further clinical studies to elucidate the in vivo relevance in metformin-treated patients regarding drug disposition and/or pharmacodynamic consequences. The unchecked proliferation and invasion typical of cancer cells are processes that can only be sustained when there is sufficient energy supply, a feature that indicates the occurrence in transformed cells of distinct phenotypes that necessarily involve elements of the intermediary metabolism. In solid tumors it has been shown by Otto Warburg that cells have adapted to rely on anaerobic glycolysis as a strategy to maintain their prevailing anabolic status. However, the upregulation of glycolysis exhibited by cancer cells does not necessarily imply a strict anaerobic phenotype nor a dysfunctional oxidative phosphorylation system. Rather, it is believed that the normal interplay between the glycolysis in the cytosol and OXPHOS in the mitochondria becomes disturbed or reprogrammed in tumor cells. The Crabtree effect observed in cancer cells, or in rapidly proliferating cells exemplifies the intimate connection between glycolysis and the oxidative metabolism. Interestingly, the anaerobic phenotype exhibited by cancer cells may in 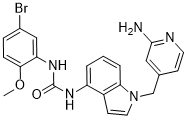 fact represent the cause rather than the consequence of the adaptive pressure. By considering that the glycolytic switch typical of cancer cells is acquired at the very onset of carcinogenesis, the idea arose that alterations in the glycolytic pathway may predispose cells to malignant transformation. Selective advantages for the transformed cells could result from various features. For instance, it is known that hypoxia-inducible factor-1 greatly stimulates the expression of glucose and monocarboxylate transporters, glycolytic enzymes and induces a down regulation in pyruvate dehydrogenase complex.
fact represent the cause rather than the consequence of the adaptive pressure. By considering that the glycolytic switch typical of cancer cells is acquired at the very onset of carcinogenesis, the idea arose that alterations in the glycolytic pathway may predispose cells to malignant transformation. Selective advantages for the transformed cells could result from various features. For instance, it is known that hypoxia-inducible factor-1 greatly stimulates the expression of glucose and monocarboxylate transporters, glycolytic enzymes and induces a down regulation in pyruvate dehydrogenase complex.
Bortezomib and paclitaxel alone did not significantly affect the total levels and phosphorylation of Bcr-Abl T315I
An excellent response with a complete remission, maintained for more than 4 years since the patient’s initial diagnosis and beginning of the treatment was observed. Based on these results, more than five different clinical trials have been initiated, using bortezomib alone or in combination with other drugs for the treatment of CML and/or Ph+ALL. Thus, bortezomib is a promising treatment in Bcr-Abl-positive leukemias. An interesting study suggested that bortezomib in combination with the cyclin-dependent kinase inhibitor flavopiridol synergizes to induce apoptosis in CML cells. Flavopiridol causes an inhibition of the cell cycle in G1 or G2, based on the inhibition of CDK. Other studies have shown that INCB28060 side effects leukemic cells are 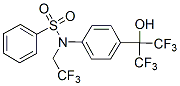 particularly sensitive when survival pathway inhibitors are combined with mitotic inhibitors. Moreover, combination of bortezomib with mitotic inhibitors are currently in clinical trials for the treatment of non-small-cell lung carcinoma and other solid tumors. Thus, we hypothesized that a strategy based on the combined treatment with bortezomib and mitotic inhibitors for the treatment of Bcr-Abl-positive leukemias may be promising. Especially important might be to determine the effectiveness of this strategy in TKIs-resistant Bcr-Abl-positive cases. Paclitaxel, a mitotic inhibitor drug acting by stabilization of microtubules, is FDA approved for the treatment of lung, ovarian, breast cancers and advanced forms of Kaposi’s sarcoma. Paclitaxel is now in clinical trials for the treatment of CML. However, to our knowledge, there are no clinical trials or published studies employing the combined bortezomib and paclitaxel regimen for the treatment of Bcr-Ablpositive CML. Such a combination, if synergistic in inducing apoptosis in Bcr-Abl-positive cells, would significantly decrease the dose of each compound necessary to achieve a therapeutic effect. Here we demonstrate that bortezomib, in combination with the mitotic inhibitor paclitaxel, efficiently kill TKIs-resistant and sensitive Bcr-Abl-positive leukemic cells. In addition, bortezomib in combination with either paclitaxel or BI 2536, another mitotic inhibitor that inhibits PLK1, induces a marked downregulation of total and phosphorylated Bcr-Abl protein levels, thus downregulating the critical Bcr-Abl downstream signaling pathways and activating caspases. Similarly, bortezomib, in combination with other mitotic inhibitors, is able to decrease Bcr-Abl activity and increase caspase activation. Taken together, our findings unravel a novel promising treatment for TKIs-resistant and sensitive CML cases, as well as other Bcr-Abl positive leukemias. The effects of 9nM bortezomib, 6nM paclitaxel or both drugs in combination were evaluated in both parental K562 and TKIs-resistant K562-R cells after 48h of treatment. Similar to K562, K562-R cells are synergistically killed by bortezomib/paclitaxel regimen, as shown by a CI lower than 1. Notably, combined treatment with bortezomib and paclitaxel strongly decreases phosphorylation of Bcr-Abl, in both K562 and K562-R cell lines. The effects of 4nM bortezomib, 5nM paclitaxel or both drugs in combination for 48h were also evaluated in both parental LAMA84 and TKIs-resistant LAMA84-R cells. Interestingly, the LAMA84-R cells show a significant increase in total levels and phosphorylation of Bcr-Abl oncoprotein when compared with parental LAMA84 cells. This suggests that these cells adapted to resist imatinib, dasatinib and nilotinib treatments through the upregulation of Bcr-Abl levels and activity. Combined treatment with bortezomib and paclitaxel was able to downregulate total levels and phosphorylation of Bcr-Abl in LAMA84-R cell lines. The T315I point mutation in Bcr-Abl is known to confer resistance to imatinib, dasatinib, nilotinib, and bosutinib. It is XL880 849217-64-7 therefore important to test whether the combined treatment of bortezomib and paclitaxel is also active on cells expressing Bcr-Abl with the T315I mutation.
particularly sensitive when survival pathway inhibitors are combined with mitotic inhibitors. Moreover, combination of bortezomib with mitotic inhibitors are currently in clinical trials for the treatment of non-small-cell lung carcinoma and other solid tumors. Thus, we hypothesized that a strategy based on the combined treatment with bortezomib and mitotic inhibitors for the treatment of Bcr-Abl-positive leukemias may be promising. Especially important might be to determine the effectiveness of this strategy in TKIs-resistant Bcr-Abl-positive cases. Paclitaxel, a mitotic inhibitor drug acting by stabilization of microtubules, is FDA approved for the treatment of lung, ovarian, breast cancers and advanced forms of Kaposi’s sarcoma. Paclitaxel is now in clinical trials for the treatment of CML. However, to our knowledge, there are no clinical trials or published studies employing the combined bortezomib and paclitaxel regimen for the treatment of Bcr-Ablpositive CML. Such a combination, if synergistic in inducing apoptosis in Bcr-Abl-positive cells, would significantly decrease the dose of each compound necessary to achieve a therapeutic effect. Here we demonstrate that bortezomib, in combination with the mitotic inhibitor paclitaxel, efficiently kill TKIs-resistant and sensitive Bcr-Abl-positive leukemic cells. In addition, bortezomib in combination with either paclitaxel or BI 2536, another mitotic inhibitor that inhibits PLK1, induces a marked downregulation of total and phosphorylated Bcr-Abl protein levels, thus downregulating the critical Bcr-Abl downstream signaling pathways and activating caspases. Similarly, bortezomib, in combination with other mitotic inhibitors, is able to decrease Bcr-Abl activity and increase caspase activation. Taken together, our findings unravel a novel promising treatment for TKIs-resistant and sensitive CML cases, as well as other Bcr-Abl positive leukemias. The effects of 9nM bortezomib, 6nM paclitaxel or both drugs in combination were evaluated in both parental K562 and TKIs-resistant K562-R cells after 48h of treatment. Similar to K562, K562-R cells are synergistically killed by bortezomib/paclitaxel regimen, as shown by a CI lower than 1. Notably, combined treatment with bortezomib and paclitaxel strongly decreases phosphorylation of Bcr-Abl, in both K562 and K562-R cell lines. The effects of 4nM bortezomib, 5nM paclitaxel or both drugs in combination for 48h were also evaluated in both parental LAMA84 and TKIs-resistant LAMA84-R cells. Interestingly, the LAMA84-R cells show a significant increase in total levels and phosphorylation of Bcr-Abl oncoprotein when compared with parental LAMA84 cells. This suggests that these cells adapted to resist imatinib, dasatinib and nilotinib treatments through the upregulation of Bcr-Abl levels and activity. Combined treatment with bortezomib and paclitaxel was able to downregulate total levels and phosphorylation of Bcr-Abl in LAMA84-R cell lines. The T315I point mutation in Bcr-Abl is known to confer resistance to imatinib, dasatinib, nilotinib, and bosutinib. It is XL880 849217-64-7 therefore important to test whether the combined treatment of bortezomib and paclitaxel is also active on cells expressing Bcr-Abl with the T315I mutation.
We could not detect any binding of recombinant to phospholipids whereas responsible for substrate specificity
Activation of trypsinogen is an obligatory step in the pathogenesis of acute necrotizing pancreatitis. So far, it is not fully understood how trypsinogen is activated prematurely in vivo. This function might be executed intracellularly by cathepsin B. Some authors also suggest a role of EP by reflux of duodenal fluid into the pancreatic duct. Ginsenoside-Rg3.gif) However, this theory remains controversial. Outside the digestive system, EP and its substrate trypsinogen are present in keratinocytes during their terminal differentiation and might be involved in the regulation of desquamation. They are also expressed by oral squamous cell carcinoma and prostate cancer cell lines. Active trypsin can increase tumor cell invasiveness. Regulation of EP by protease inhibitors may therefore be not only important in the digestive system, but also in epidermal differentiation and tumor invasion. It was the first time that an interaction of EP with a serpin-type inhibitor was shown. Additionally, it was also the first time that PD325901 inhibition rate constants and the stoichiometry of inhibition were calculated for the interaction of a transmembrane serine protease with PCI. It has been shown previously that heparin and phospholipids are able to stimulate or to reduce the GDC-0879 Raf inhibitor inhibitory activity of PCI towards several proteases. Glycosaminoglycans like heparin seem to regulate the inhibitory activity of PCI by binding to the target protease as well as to the serpin. In case of PCI, this bridging mechanism is strongly protease-dependent and often leads to enhancement of protease inhibition. Interestingly, the inhibition of plasma kallikrein by PCI is not stimulated by heparin, factor Xa inhibition shows only a slight stimulation, and the interaction of PCI with tissue kallikrein is completely abolished in the presence of glycosaminoglycans. Heparin slightly reduced the inhibition of recombinant human EP by PCI and this effect was even more pronounced using bovine EP purified from calf intestine. This could be explained by the fact that the recombinant EP carries a positively charged His-tag at the Cterminus which might counteract the repulsive effect of the negatively charged heparin. AT, on the other hand, inhibited EP only when heparin was present. So, heparin stimulated the inhibition of EP by AT, but reduced the inhibition of EP by PCI. To our knowledge, this is the first demonstration that heparin led to a reduced inhibition of a particular protease by PCI, but an increased inhibition by AT. This may be due to differences in regulation of serpin activity by heparin, as AT undergoes a conformational change when bound to glycosaminoglycans compared to the bridging mechanism of PCI. Furthermore the different heparin-binding sites of PCI and AT may also contribute to this opposed effect. As mentioned above, native EP is a type II transmembrane serine protease. It contains an N-terminal hydrophobic segment from position 18 to 44, predicted to span the membrane. The recombinant EP used is a mixture of two forms, in which the heavy chain is truncated and starts either at Leu41 or Ser118. Nterminal sequence analysis by Edman degradation revealed that also the bEK contains a mixture of two heavy chains starting at Gly53 and Ser118 respectively. Phospholipids did not influence EP inhibition by PCI. Assuming a heparinlike bridging mechanism for the stimulatory effect of phospholipids on PCI-protease interactions, these results are not surprising, since it has been shown previously that a truncated EP lacking the transmembrane domain does not interact with phospholipid vesicles. Supporting this data, a commercially available protein-lipid overlay assay containing membrane phospholipids was performed.
However, this theory remains controversial. Outside the digestive system, EP and its substrate trypsinogen are present in keratinocytes during their terminal differentiation and might be involved in the regulation of desquamation. They are also expressed by oral squamous cell carcinoma and prostate cancer cell lines. Active trypsin can increase tumor cell invasiveness. Regulation of EP by protease inhibitors may therefore be not only important in the digestive system, but also in epidermal differentiation and tumor invasion. It was the first time that an interaction of EP with a serpin-type inhibitor was shown. Additionally, it was also the first time that PD325901 inhibition rate constants and the stoichiometry of inhibition were calculated for the interaction of a transmembrane serine protease with PCI. It has been shown previously that heparin and phospholipids are able to stimulate or to reduce the GDC-0879 Raf inhibitor inhibitory activity of PCI towards several proteases. Glycosaminoglycans like heparin seem to regulate the inhibitory activity of PCI by binding to the target protease as well as to the serpin. In case of PCI, this bridging mechanism is strongly protease-dependent and often leads to enhancement of protease inhibition. Interestingly, the inhibition of plasma kallikrein by PCI is not stimulated by heparin, factor Xa inhibition shows only a slight stimulation, and the interaction of PCI with tissue kallikrein is completely abolished in the presence of glycosaminoglycans. Heparin slightly reduced the inhibition of recombinant human EP by PCI and this effect was even more pronounced using bovine EP purified from calf intestine. This could be explained by the fact that the recombinant EP carries a positively charged His-tag at the Cterminus which might counteract the repulsive effect of the negatively charged heparin. AT, on the other hand, inhibited EP only when heparin was present. So, heparin stimulated the inhibition of EP by AT, but reduced the inhibition of EP by PCI. To our knowledge, this is the first demonstration that heparin led to a reduced inhibition of a particular protease by PCI, but an increased inhibition by AT. This may be due to differences in regulation of serpin activity by heparin, as AT undergoes a conformational change when bound to glycosaminoglycans compared to the bridging mechanism of PCI. Furthermore the different heparin-binding sites of PCI and AT may also contribute to this opposed effect. As mentioned above, native EP is a type II transmembrane serine protease. It contains an N-terminal hydrophobic segment from position 18 to 44, predicted to span the membrane. The recombinant EP used is a mixture of two forms, in which the heavy chain is truncated and starts either at Leu41 or Ser118. Nterminal sequence analysis by Edman degradation revealed that also the bEK contains a mixture of two heavy chains starting at Gly53 and Ser118 respectively. Phospholipids did not influence EP inhibition by PCI. Assuming a heparinlike bridging mechanism for the stimulatory effect of phospholipids on PCI-protease interactions, these results are not surprising, since it has been shown previously that a truncated EP lacking the transmembrane domain does not interact with phospholipid vesicles. Supporting this data, a commercially available protein-lipid overlay assay containing membrane phospholipids was performed.